
We all know neat little things that help us do stuff. Here are some of mine, first the analysis ones, then the making stuff ones…
- Any Resistor ! (from a set of 1, 3.3, 10, 33, 100, 330…)
- Spreadsheets, Statistics and Monte Carlo #spreadsheets
- Dimensional Analysis and Reductio Ad Absurdum #dimensional
- The Search for Simplicity and Symmetry #simplicity
- Right Angles: Euclid, Pythagoras and Nearly Pythagoras #rightangles
- Multimeters and Datalogging #multimeters
- Take-Aparts: the Free Parts Supplier ! #takeaparts
- Meccano/Erector Set Parts #meccano
- Clamp Stands #clampstands
- Finger Bowls and Hot-melt Glue #hotmeltglue
- Rubber: its Gripping Stuff #rubbergrip
- Oil: its anti-Gripping Stuff #oillubricant
- Grease: more anti-Gripping Stuff #grease
- Ball-bearings: the World goes round on ball-bearings #ballbearings
- Left Hand Gloves #lefthandgloves
- Soldering Wires and Strain Relief #strainreliefwires
- NiMH (and other) Batteries #nimhbatteries
- Variable Power Supplies and Diode Power Reduction #variablepower
Now click on the links to take a look at some of the details on these… NB, still under construction in places…
…
Any resistor
You may find yourself short of a resistor of the right value for part of a project. Nil desperandum ! The solution is simply a pair, or very occasionally a triple, of resistors in series or parallel. The following shows how:
| R values | R values | |||
| 1.00 | 3.63 | |||
| 1.10 | 4.30 | |||
| 1.33 | 5.00 | |||
| 1.65 | 5.50 | triple | (3.3+3.3) par 33 | |
| 2.00 | 6.00 | triple | (10 par 10) + 1 | |
| 2.48 | 6.60 | |||
| 3.00 | 7.67 | |||
| 3.30 | 9.09 | |||
| 10.00 | ||||
| R | Rcombo | Series | ||
| 0.1+ | 0.33+ | 1+ | 3.3+ | |
| 0.10 | 0.20 | |||
| 0.33 | 0.43 | 0.66 | ||
| 1.00 | 1.10 | 1.33 | 2.00 | |
| 3.30 | 3.40 | 3.63 | 4.30 | 6.60 |
| 10.00 | ||||
| R | Rcombo | Parallel | ||
| 3.3 PAR | 10 PAR | |||
| 3.30 | 1.65 | |||
| 10.00 | 2.48 | 5.00 | ||
| 33.00 | 3.00 | 7.67 | ||
| 100.00 | 3.19 | 9.09 |
Spreadsheets, Statistics & the Monte Carlo Method
Spreadsheets, that computer application that finance people love, is also just fine and dandy for doing all sorts of science analysis. But buried inside these financially useful things like adding up and averaging and plotting graphs versus time are all the things you get on a scientific calculator and more, much more, stuff that is really useful in science and experiments. Things like log-log plots, frequency histograms and random numbers.
And to crown it all, if you really need it, behind every square on the spreadsheet you can put a computer program ! As well as ‘macros’, simple recording of keystroke programs, there is real BASIC programming too.
There are lots of statistics functions, averages and standard deviations, which are great to make sense of numbers coming in from experiments especially the flood of numbers that can come in from datalogging (see Datalogging below). Other features for dealing with large number sets include re-ordering sets of numbers, smoothing filters, Fourier analysis (eg. looking for the frequency spectrum of a complex audio waveform), correlation and frequency histograms.
The Correlation Trap: Correlation is often an aid to understanding: if measurement A correlates with result B, then you are half-way to proving that A causes B. But beware: correlations are the statistic that has launched a thousand shiploads of scientific papers which were subsequently proved wrong. Correlation is NOT causation, it only suggests its. Even if you have a correlation coefficient coefficient of 0.99 and a scatter of points in X-Y lying almost straight along a line, you may have coincidence, not cause.
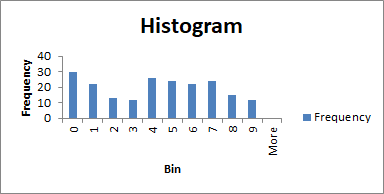
While a frequency histogram is just a fancy name for a graph where you plot the number of times a number comes up versus what that number is. They are really helpful in studies where random variations are concerned, which brings us to another thing.
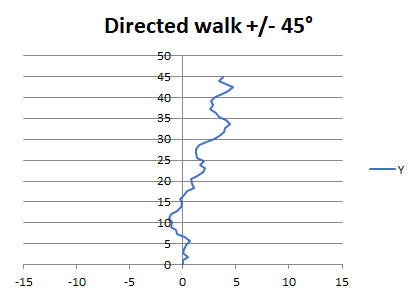
It often happens that something that you don’t fully understand, or can’t do the maths for, can be analyzed by the curious approach of simulating it, using random numbers as input, and then looking at the results and comparing to experiments. This is, in essence, what the Monte Carlo Method is about. Take, for example, navigation using a compass at sea. How good does a compass need to be, to go efficiently from A to B, if we assume that B is a long coastline where it doesn’t matter too much where you make landfall. Here is a graph I came up with for the light-polarization-based ‘Viking compass’. It plots the slightly drunken wandering of a boat with a compass that varies randomly from -45 to +45 degrees from its course. Even with such a bad compass, as this plot shows, you can still get places ! (see Optical Realities on this website).
‘Doing the Numbers’
Finally, it is always worth few minutes putting your formula into a spreadsheet and putting some real input numbers in to see what it comes up. You may be surprised at how many ‘bloopers’ this throws up. Even if you don’t actually use a spreadsheet, ‘doing the numbers’ – working out a numerical example of some algebraic formula you have developed – is enormously useful. And don’t forget to check the decimal point: you can often live with being a few % out on something. But you don’t want to be a factor of 10 wrong !
Orthophilia: Love of Straight Lines: Getting a set of data to the point where you can draw a straight line graph is a powerful indicator that you really understand something. Spreadsheets are a powerful means of satisfying your Orthophilia. Try a simple plot. Not straight ? Some spreadsheet magic may help. Try a log-log plot. If the slope is β, then you may have a y = m x^β + c relation. Try a log-linear plot. It could be you have an exponential phenomenon like radioactive decay. Try other things – try ‘modelling’ – substituting a formula y = f(x), plot that versus your data and x… Finally, with luck, you get a straight line, y = .x + c ! Eurkea ! Celebrate ! Nibble a chocolate bar ! Sip a coffee ! Or whatever takes your fancy ! You’ve probably understood the science.
Calculus: curves, rates of change and areas from spreadsheets: More power from your calculus – but it is limited ! One of the wonderful things that became possible in the 1970s was the use of a computer to solve the unsolvable – to do with massive computation what had before then to be done with pure ingenuity in maths. The spreadsheet puts this power at the fingertips of everyone !
…
…
The Search for Simplicity and Symmetry
Simplicity is one of principles of all science: it is sometime caused Occam’s Razor, and it has been around at least as far back as William of Ockam
(1287 – 1347). Occam simply says that the simplest explanation that fits all the facts. If you found a small rodent-toothed four-legged vertebrate skeleton on the Moon, the simplest explanation is that a mouse stowed away on a moon rocket. It is possible, but rather less likely, that the Moon had life, developed bacteria, then algae, plants, fish, insects, and finally land mammals, and that this evolution followed an almost identical course as that on Earth and led to a mouse-like mammal.
Symmetry is in some ways part of that search for simplicity. Explode gunpowder between two similar ball bearings and one will go in the opposite direction at the same speed as the other. Throw something up on the Moon and it will follow a parabolic trajectory: the same shape up and down: symmetrical around the zenith. In each case you only need to calculate one half of the problem. Or if you do a calculation in which the solution to the two halves of the problem look different, then look out ! – maybe you have made a mistake.
It is not simply about symmetry of shape. Symmetry in the Laws of Nature, for example, demand that something that happens on (or near) Planet Earth can happen in exactly the same way on or near Planet Ogzinablatt orbiting a star in the Andromeda Nebula. There is a symmetry in time as well as in place. If it happens now, then it has happened in the same way in the past and will happen in the same way in the future: that’s the foundation of the science of geology, inter alia. It is even the case that time can be reversed in most microscopic processes in physics. Time symmetry breaks down only when thinking about the random motions of huge numbers of atoms or molecules: at the macroscale they will move in the direction of entropy – greater disorder – even though at the microscale their interactions are perfectly reversible.
…β
…
…
Dimensional Analysis and Reductio Ad Absurdum
Often, people are familiar with Dimensional Analysis, or The Method Of Dimensions, but they then don’t remember to use it. Other people are vaguely aware of it because they know that the units of something must be right. If you don’t know about it, or need a refresher, then look it up. But here is a brief reminder: A simple example of dimensional analysis is provided by Newton’s acceleration equation F = ma, which relates force applied F to mass m and acceleration a. The dimensions of mass are simply M, for mass, while acceleration, the rate of change with time T of velocity v, which itself is the rate of change of distance L with time T, has dimensions L T¯ ¹ , so acceleration is V T¯ ¹, giving you L T ¯ ² . We can now find the dimensions of force: it must be those of m.a , in other words M L T ¯ ². We can now use this to calculate the dimensions of energy. Since energy E can be measured by force x distance, we have E has dimensions M L² T ¯ ². We can immediately see that the formula E = ½ m v² for kinetic energy has correct dimensions.
In the same way that you can use Dimensional Analysis to make simple checks based on the units, you can use ‘reductio ad absurdum’ to check out that at extremes your analysis, the algebra you have done, works at ‘absurd’ extremes. For example, suppose you fit your data measurements Y versus time T to an exponential formula like :
Y= exp(aT)
where a is a constant. If a turns out to be a positive number, then your analysis is most likely flawed. Why ? Because if you look at what the formula does when T is large, you will see that Y becomes insanely – one might say absurdly ! – large.
Finally, if you are still uncertain whether your analysis is correct, ‘sleep on it’. Wake up the next morning and take another look – you will be surprised how often a simple mistake glares back at you from the page ! I have tested how well ‘sleeping on it’ works with cryptic crosswords. With a tricky crossword I often end up one evening at a dead end with half-dozen fiendishly difficult conundrums. The next morning it all looks so much easier, and the solutions just pop up.
…
…
…
Right Angles: Euclid, Pythagoras and Nearly Pythagoras
It is often the case that you will need a right angle in something. It could be anything from a piece of wood to a metal frame construction. Now you can often deploy that mainstay of the carpenter, the set-square, or its paper-drawing equivalent. But it is often more accurate to use a ruler and compass method, just as the ancient Greeks did 2500 years ago. Just draw up a equilateral triangle with the compass, which gets you 60° angles, now divide one of those into two to get 30° and add the adjacent 60° and Bingo ! you have your right angle. There are other ways of doing it, of course – just playing around with ruler and compass and you’ll stumble on them. Other angles are available of course. You can probably figure how the ancient Greeks calculated square roots using a ruler and compass by playing around too and remembering Pythagoras, although you may finding a cube root trickier (but yes, it IS possible).
But that’s not the only old Greek maths that you might find useful. If you are a frequency user of struts with regular holes in them, like Meccano/ Erector, or Dexion and similar shelving metal struts, then a little piece of Pythagoras can be useful. ‘I can use a 3 4 5 triangle !’, you exclaim, and indeed you can, because 3² + 4² = 5² , so these lengths form a right angled triangle. But you may want to use a bigger triangle. So use 6 8 10, or 9, 12, 15. But you may also want to use a triangle that has equal x and y arms. So use er… Trickier. You really need a 1:1:√2 triangle (that’s 1.4142). Handier numbers are 5:5:7, which approximates √2 as 7/5 or 1.4, or a 12:12:17 triangle, which approximates 17/12 or 1.4167. The former gets you an angle of 88.5°, while the latter get so close you really won’t need it better, just 0.2 degrees away from 90° .
Finally, moving away from right angles, you may find it very useful to cut an octagon, rather than trying to cut a circle. An octagon is conveniently close to a circle for most purposes, and can often be sandpapered to a circle with very little wasted material, or put on a lathe. But how to draw an octagon on a given square of material from the wood yard ? Its all down to Pythagoras again. Divide the side of the square by (1 + √2) or 2.4142 and you have the sides of your shiny new octagon, which you can divide by wo and put in the middle of the sides of the square. or measure in from the corners by side of square divided by (√2 (1 + √2) ) or 3.413.
…
…
…
Multimeters and Datalogging
With a simple multimeter you can measure the world ! As well as the numerous scales covering everything from the usual voltage, current and resistance, you will often find frequency and capacitance. But these are only the electrical readouts. There is a whole industry dedicated to making sensors that convert something like temperature, pressure, force, light and a host of other things into an electrical signal which you can then measure with your multimeter.

The datalogger function is also a feature of many multimeters, and this further extends their capability. A USB or similar lead allows you to suck measurements in to simple software like Teraterm ‘terminal emulator’, from which data can be put in a spreadsheet and analyzed. Finally, don’t forget that many small ‘credit card’ size computers like the Microbit (see Microbit projects section) which have analog inputs and/or sensors which can be used for datalogging. Unlike the multimeter, they can log multiple channels. For example, the Microbit has three which you can plug in a 4mm plug or clip a croc /alligator clip onto and half a dozen more you can use if you have a suitable edge connector. These computers are small enough to include on-board mobile projects – will collect and transmit data, either by USB lead or by Bluetooth radio comms.
…
…
…
Take-Aparts: the Free Parts Supplier !
As you may already be aware from projects on the Saturday Science website or the books, there are a host of useful things to be extracted from things that normally get trashed that we can use again in projects. Hair driers that blow but don’t get hot – vacuum cleaners that won’t suck – printers that don’t print – they can all be useful. Hair driers have a low voltage and power electric fan inside, while vacuum cleaners include a rotating roller brush with good bearings, flexible hose and other goodies.

Printers come with a whole bunch of gearwheels, belt drives, small versions of lab ‘light gates’, DC motors, stepper motors, bearings, rollers, and a panel of electronic parts that you just might find handy sometime, from power transistors to capacitors and diodes. And never throw the DC or AC low voltage power supply that came with any gadget that you are throwing on the scrapheap. Mostly these will work fine to power a project using a motor or electronics – see below for how to adapt them.
And finally, the bonus: with taking apart these gadgets, you get to learn about about how they work, not just the basics, but the details, what materials are used, hidden parts, all the ingenious tricks and dodges that the makers have designed into them. (Note: to avoid any risk of electric shock, remove fuse from the mains plug and cut off the mains leads before dismantling mains-powered electrical appliances).
…
…
…
Meccano/Erector Set Parts
There are other brands of these kind of parts, and larger scale variants such as Dexion, sold mostly to make specialized shelving but used for bazillions of other purposes. They are particularly useful when combined with other materials like wood, plastic etc.

…
…
…
Clamp Stands
Not just for chemistry, these gadgets are just great for so many things. During the construction of something, they can be your third (or fourth, or fifth) hand. They come in miniature too, helpful for really small assemblies and soldering. While they offer the possibility of testing something out with the minimum of construction, holding things together roughly for a first trial. If you get a lot of these clamps and stands, and spare rods and bosses, you can set up a whole experimental rig with few other parts needed.

…
…
…
Finger Bowls and Hot-melt ‘glue gun’ Glue

Hot-melt glue is great stuff for quickly assembling things. But don’t sizzle your fingers ! Keep a little bowl or tumbler of cold water near to the glue gun. If you get some on your fingers, plunge them into the water immediately and walk over to a tap, keeping the fingers in the bowl of water until you get them under the running water of the tap, where you keep them for at least a couple of minutes. There other snags about using hot-melt glue, such as the problem of gluing large areas – the start of the glue line is beginning to solidify by the time you get to the end. You have to think your way around this a little. One way is to use large blobs of glue here and there around the area to be glued, not continuous long thin lines. These stay hot longer and can be squeezed to spread out when you push the parts together. Talking of pushing parts together, it is often useful to arrange for parts to be clamped together by a vice, a ‘workmate’ type of trestle table/vice or a series of G-cramps while the adhesive sets – especially useful with water-based (e.g PVA) wood glue.
…
…
…
Rubber: it’s Gripping Stuff

Put on a pair of rubber gloves and you immediately take on the superpower of SuperGrip. SuperGripMan or SuperGripWoman can instantly open jars with lids that ordinary mortals can’t remove.
To see what is going on, you need to use the laws of friction, which mean that the force stopping something sliding is equal to the coefficient of friction times the force pushing the sliding surfaces together, the ‘normal’ force. The area of the surfaces in contact doesn’t matter, but the speed of sliding does. When stationary, the static coefficient of friction applies, which is higher than when sliding is happening, when a lower dynamic coefficient applies. As speeds rise, the dynamic coefficient falls a little. And a note of caution: all coefficients of friction vary enormously with surface condition – the presence of oxidation on metals, for example, or, (of course) lubricant, makes a huge difference. In the case of rubber gloves, it seems that SuperGripMan or SuperGripWoman can get a force at least twice what you can get with human hands alone and often 4x more, which fits with observations of static coefficients on rubber on steel of 2 or more.
And don’t forget that there are other grippy things. Rosin is a kind of anti-oil or anti-grease. Put rosin on something and its coefficient of friction. And don’t forget that there are other grippy things. Rosin is a kind of anti-oil or anti-grease. Put rosin on something and its coefficient of friction goes up from the typical range of 0.2 to 0.5 to a surprising 0.8, even up to 1 or more. Rosin is a mixture of similar organic acid, based on a -COOH group attached to three carbon rings, which forms a thin film over surfaces easily, when rubbed on them. Once on, the coated surface gets highly frictional, like the bow of a violin, which is one of the common applications for rosin. This is s complex subject: take a look at this Cambridge study… http://www2.eng.cam.ac.uk/~jw12/JW%20PDFs/Jonathan_rosin.pdf
…
…
…
Oil: it’s anti-Gripping Stuff
The world wouldn’t go round, except for oil, or at least, the industrial world. It really wouldn’t. And I don’t mean crude oil as a source of energy, I mean lubricating oil. Cars wouldn’t drive, planes wouldn’t fly, most compressors wouldn’t compress, most vacuum pumps wouldn’t pump vacuum, and most holes wouldn’t get drilled. Oil reduces friction, a lot. It also has side-benefits which are often unappreciated. It has a cooling effect. Look at truck or a car, and you will find a ‘radiator’, a panel of tiny tubes that transfer heat from the water cooling circuit to the air. (The ‘radiator’ is actually a convector, of course). But look again at the truck, or a powerful car, and you will often find a second radiator, one that just cools the oil. Oil lubricates the to and fro motion of the pistons in the cylinders, but is also heated up in the fierce heat of the fuel-air explosions in the cylinders. Cooling it helps stop the engine getting too hot. Its the same with drilling a hole. Add oil (or water, or even (not hygenic this) spittle) and a drill will drill better. Its partly lubrication, but it is also keeping the tiny cutting edge of the drill cool, so it cuts and stays sharp rather than getting so hot that it softens and gets blunted. You can drill a few holes, its true, without oil or water, but drill a lot of holes and you will find yourself with blunt drills which even if they do drill do so much slower. Much better to oil as you drill.
…
…
…
Grease: more anti-Gripping Stuff
Don’t forget to grease wood screws: making things in softwoods, as we generally do, with a touch of grease on the screws, you won’t need a pilot hole, leastways in the underneath part. Its still good to put a hole in the top part, but that often ain’t necessary either if it isn’t too thick, but only provided you grease the screw. Grease is great on lots of other things where ultra-low friction isn’t required, where you wouldn’t use oil in case it would drip out, or things with a lot of force on them. Finally, ‘low film strength’ grease, for which, read ‘Vaseline’ or ‘petroleum jelly’, is great for protecting electrical contacts such as screw terminals and battery boxes from intermittent faults caused by metal oxidation.
…
…
…
Ball-bearings: the world goes round on ball-bearings
The humble ball-bearings, and its colleagues, the roller bearings and similar, are essential but hidden parts of the modern world. They are also enormously valuable in creating frictionless pivots that science projects often require.
The most useful ball-bearing for many projects will be the general purpose deep groove radial ball-bearing, which goes on a shaft. The axial ball-bearing is like the bearing in a ‘Lazy Susan’ table roundabout platter, which goes between two surfaces.
One dodge that is especially useful for science projects is to take a commercial ball-bearing and remove the grease. Commercial bearings are designed for heavy loads, and relatively low speeds, whereas science projects often need high speed but don’t impose much load on the bearing. Removing one or both of the cover plates to reveal the balls can usually be accomplished with a tiny screwdriver. Then soaking in some white spirit a couple of times will usually do the trick. If they are the right size (and they are still available – these are a toy which goes in and out of fashion), the ‘fidget spinner’ is a valuable source of bearings which are clean – they are not greased. Fidget spinners always have a small high speed bearing in the middle, but may also have 3 or more additional slightly larger bearings in the ‘arms’.

Finally, don’t forget that bikes have 10 or so ball bearings which can be redeployed: two on each wheel, two on the front forks and (most people forget this) two on each pedal.
…
…
…
Left Hand Gloves
Left hand gloves are a ‘handy’ way of protecting your left hand from your right hand and the sharp or heavy object you are wielding in your right hand. (Obviously, if you are left-handed, the single glove goes on your right hand.) A glove with rubber finish also allows you get a grip on things you are working on nicely, as discussed above.
…
…
…
Soldering Wires and Strain Relief
Its a useful skill to be able to solder together a few wires together. Hooking things together by twisting the wires together is very limited, and joining with ‘choc bloc’ screw terminal strips is better, but limited too. And there are prototyping boards available which work reasonably well up to a few dozen parts, provided the parts all have solid wire ends or are ‘dual-in-line’ microcircuit packaging. But soldering is King…
Unfortunately, soldering is a process designed especially for the intelligent Octopusoid creatures who inhabit the outer planets of Betelgeuse. All you need to do is hold wire or part A, wire/part B, and soldering iron, and then use your fourth hand to apply the solder. Parts of the process, it is true, have gotten easier. Electronic solder wire is now almost 100% tin, free of the percentage of lead it used to have. And it includes a few% flux, which will clean the metal slightly and allow the solder to flow more freely onto the wires, although they should still be very clean, bright metal. Helpful too is that wires and connecting tags on components are now invariably tin-plated, rather than plain copper. Flux is handy too. Keep a little flux in a tin handy for soldering things that the molten solder won’t wet. There is a handy flux and solder made for aluminium which works on steel and NiChrome resistance wire. Try to anchor parts and wires in some way, so that you can concentrate on the solder and soldering iron. A miniature clamp stand, small pieces of ‘Blutak’ can be pressed into service. For more than half a dozen parts and wires, use a ‘stripboard’, with its rows of holes to insert small components.
Finally, we come to the problem of wires pulling off parts and circuit boards. Try wiggling your freshly-soldered wire to and fro a few times. I bet that within a dozen wiggles that wire will snap off near to the solder blob at the end. Of course you can be careful not to let wiggling of wires happen much, but it is almost impossible to eliminate entirely. The solution is strain relief. Clamping the wires is one way out. Or taping them, or encapsulating them in a blob of hot-melt glue up to a few millimetres above the insulation starts.
Now try wiggling your freshly-soldered wire to and fro a few times. I bet that within a dozen wiggles that wire will snap off near to the solder blob at the end. Of course you can be careful not to let wiggling of wires happen much, but it is almost impossible to eliminate entirely. The solution is strain relief. Clamping the wires is one way out. Or taping them, or encapsulating them in a blob of gluegun glue up to a few millimeters above the insulation starts.
…
…
…
NiMH (and other) Batteries

NiMH (and other) Batteries
Nickel Metal Hydride Rechargeable cells give 1.25 volts or so, can be recharged up to 500x or so. So a $1 AA cell can do the service of around 125 25 cent alkaline AA cells, with a saving of $30 ! They can give high peak currents – several amps from an AA cell – and they don’t, in contrast to NiCd cells, lose their charge over a few weeks. They do lose charge, but take months. And you don’t have to worry about leaving gear switched on by accident.
They are also robust mechanically and electrically. You can overload them by drawing too much current for a short while usually without damage and they aren’t too fussy about how they get recharged – almost any charger or around the right voltage will do. This is in contrast to lithium cells, many of which are easily damaged if recharged except with their special charger.
Alkaline Batteries.
Non-rechargeable Llthium Batteries have great performance: huge capacity, double the voltage of an alkaline cell (3V instead of 1.5) and will sit happily for 10 years before being perfectly OK ! There is a teeny-weeny disadvantage, however: they cost more than 10x as much ! And you also need to be a bit careful if you buy a standard size like AA, the doubled voltage, 3V, may blow your 1.5V circuit to bits ! (There are lower voltage lithium cells, but these seem to me to throw away some of the real advantage of lithium – its huge capacity). There are also really nice rechargeable lithium cells. However, you need to be very careful to link these to a properly regulated charger circuit. That seems to me to be extra work that you don’t need to do – unless you really need high capacity and you need rechargeability.
Zinc Carbon, and to a lesser extent Zinc Chloride batteries shouldn’t be used in projects. Their performance is poor (typically higher internal resistance, lower capacity) means that they won’t run a project for very long. These low quality cells will not keep their charge for more than a year or two – they self-discharge. And to crown it all, they have a nasty habit, particularly when old/fully discharged of leaking a slightly corrosive goo from inside their cans all over your project.
Internal Resistance is an important characteristic of a battery. An AA alkaline cell has an internal resistance of 0.2 ohms. Which means that if you connect it to a motor which draws 1A of current, 0.2Volts will be drop be dropped inside the battery, that’s just Herr Ohm’s Law V = I R. So your 1.5V volt battery will only have (1.5 – 0.2) = 1.3 volts left to power the motor. That’s probably fine. NIMH batteries have lower internal resistance, eg. 0.05 Ohms for an AA cell, which means that they would drop from 1.2 to 1.15 volts at a 1A current, which compensates to an extent for the lower basic voltage.
Finally, don’t even think of using a PP3 battery (or similar squarish looking cells – see the pic above) for running an electric motor. They are designed to give maximum currents of only 100mA, ideally much, much less. They mostly have high internal resistance, 2 ohms for alkaline, much higher for zinc cells. They can even get quite warm (because of all that power being dissipated inside them) which isn’t good for them. And they are an expensive source of power relative to the cylindrical cells. If you need high voltage, use a battery box and cylindrical cells.
Clearly internal resistance needs to be taken account of when using higher current devices like motors. There is some alleviation on this if you use a high current motor which doesn’t have much of a load on it: the current drawn might be 3A on start up, when the motor is going from stationary to operating speed, but at operating speed the motor might well draw much less current. The difference between start-up and running current grows with motor size, until huge powerful low voltage motors can consume only an amp or two when running, but 50A when starting up (model plane flight motors are like this). If you really need high currents, use cells made from lead-acid or, better, NiCd technology: these are a bit esoteric, and not high in energy content, but not particularly expensive. They can provide internal resistance in the milliohm region, and can provide tens or hundreds of amps (eg. for starting car engines). But watch out for safety problems if you short circuit these cells – wires may get hot enough to glow red and set fire to things !
…
…
…
Variable Power Supplies and Diode Power Reduction
It is often useful, sometimes essential to be able to control the level of electrical power applied to a motor or other actuator in a project. The best way is some kind of variable power supply unit (PSU). You don’t have to pay too much, these days, for a box which will give a few amps at up to 30 volts, which will be great for powering, or at least testing, a whole bunch of gadgets that you make. They usually come with built-in current and voltage meters, and even a current limiter control as well as the voltage control, all of which is great.
But including such a variable PSU in a project isn’t always best, which is where other power supplies and batteries (for things that need to move without trailing wires) come in. With batteries, for me, the NiMH is king, see above. But plug-in DC PSUs are handy and don’t need recharging. There are some with a selector switch going from, eg. 3 to 18V in 3 volt increments, which is handy. But once you have captured some PSUs from redundant equipment (see Take-Aparts above), then you may have free PSUs for projects in various voltages.
Whether battery or fixed PSU, you can often readjust the voltage supplied in a simple way: add a diode or two. Although proper electronic regulator circuits are fairly easy to use, they still need several parts to make them go, and may be subject to overheating. Nothing could be much simpler than putting a silicon diode or several diodes in series with a PSU. Each diode will take the voltage down by 0.65 Volts or so, which happily allows you to select voltages in between NiMH battery packs. Want 2.5 volts ? Thats 2x NiMH cells. 3.75 volts ? That 3x NiMH cells. 3.1 volts ? Thats 3x NiMH cells + one diode in series. Similarly, with a fixed PSU, suppose you have a 5V PSU (very common size, BTW, used for all sorts of electronics and USB powered kit of course). But you need closer to 3V. Three diodes in series gives you 5 – 3x 0.65 = 5 – 1.95 = 3.05 . Bingo ! Close enough to 3V. *
*for very small currents, the diode may not drop as much as 0.65V, so to be sure, wire a resistor than will draw a milliamp or two between the diode output and ground. Also, note that for some purposed, what you need may be a resistor, not a diode. If you just want a low torque from an electric motor, for example, just putting a resistor R in series with the PSU of voltage V will work. Electric motors have a low resistance, so the result is a current will flow that is given by I = V / R, with I pretty much independent of what the motor is doing.
LEDs, Light-emitting Diodes as Light-receiving Diodes and other electronic curiosities:
It may not occur to you, but many semiconductors are light-sensitive. Early transistors were housed in little glass capsules, which had to be painted to avoid this. Scrape the paint off and you had a phototransistor ! Similarly today, and without having to scrape paint, if you have an LED, you also have a light sensor. More coming soon…